Year-7 Chemistry Particles: Evaporation and Sublimation
In this Year-7 Chemistry Particles: Evaporation and Sublimation lesson, students will learn how to explain why liquids can evaporate below their boiling point, explain why we sweat and describe sublimation and deposition.
The slides include extension questions, and students can choose from a variety of different tasks.
This lesson includes:
- Title slide with learning objectives
- A starter activity
- A group reading task (literacy)
- Keywords sorting task (vocabulary: boiling, evaporation, condensation, sublimation and deposition)
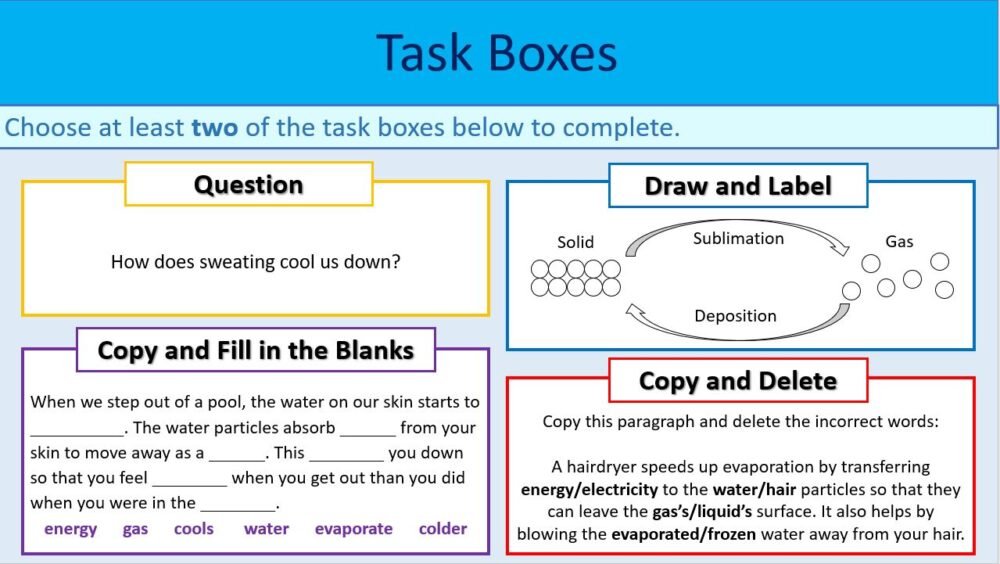
- A task box exercise
- A sorting task to summarize the difference between boiling and evaporation
- A handout sheet on state change
- Recall questions on particle theory and state change
Get this resource as part of the KS3 Chemistry: Particles Bundle Year 7 and save up to 33%
All content, diagrams and photos © Troilo Science / Febo Learning unless otherwise stated.
Next Lesson: Diffusion
In this lesson students will learn how to describe how particles spread out by diffusion, explain the factors which affect how quickly diffusion happens and give some examples of diffusion.
Lessons for Key Stage 3, Year 7 Chemistry Topic 1 Particles
Particle Model
States of Matter
Melting and Freezing
Boiling and Condensing
Evaporation and Sublimation
Diffusion
Gas Pressure
Particles End of Topic Revision